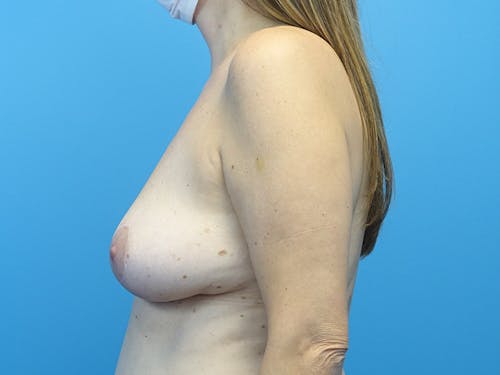
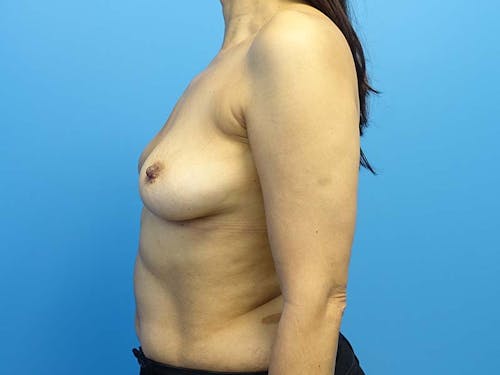
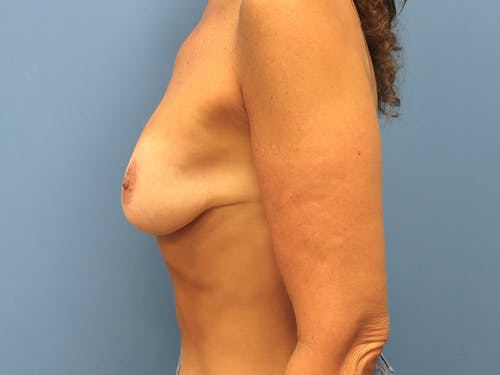
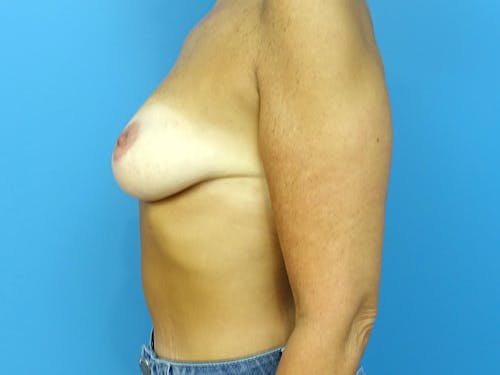
Whether for cosmetic breast augmentation or breast reconstruction, it is known that breast implants are not lifetime devices.
Breast Explant Surgery
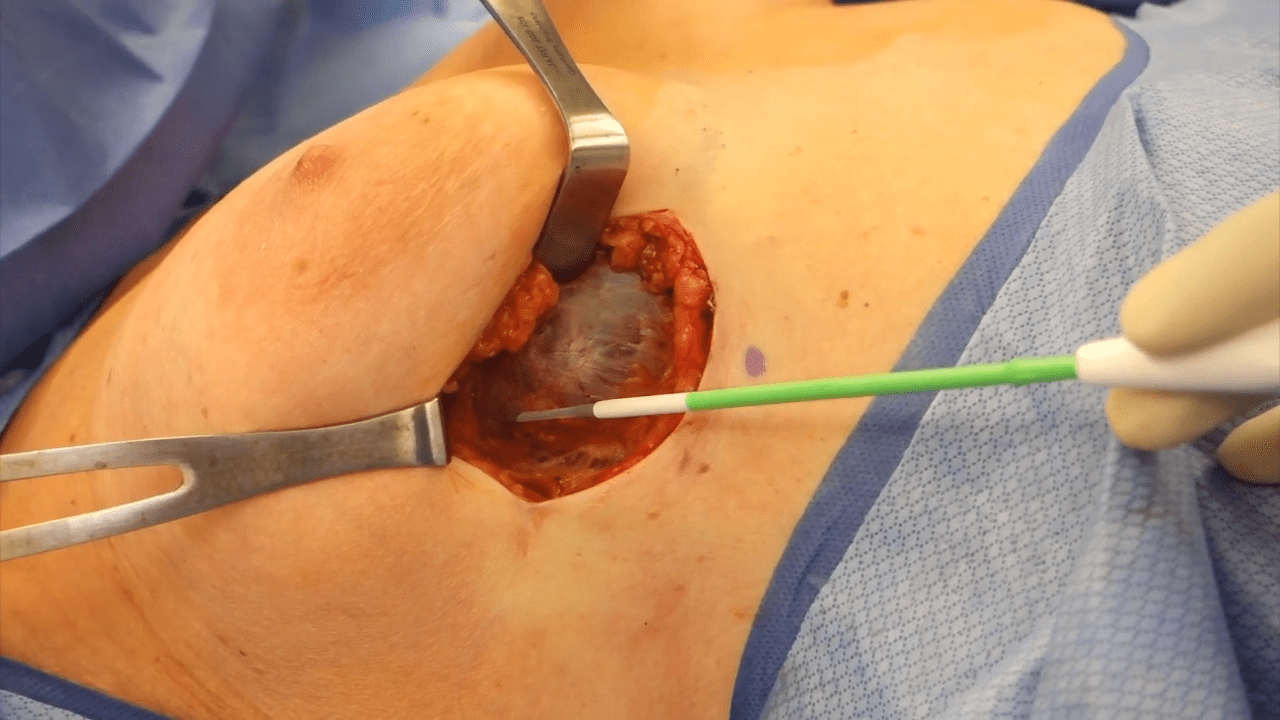
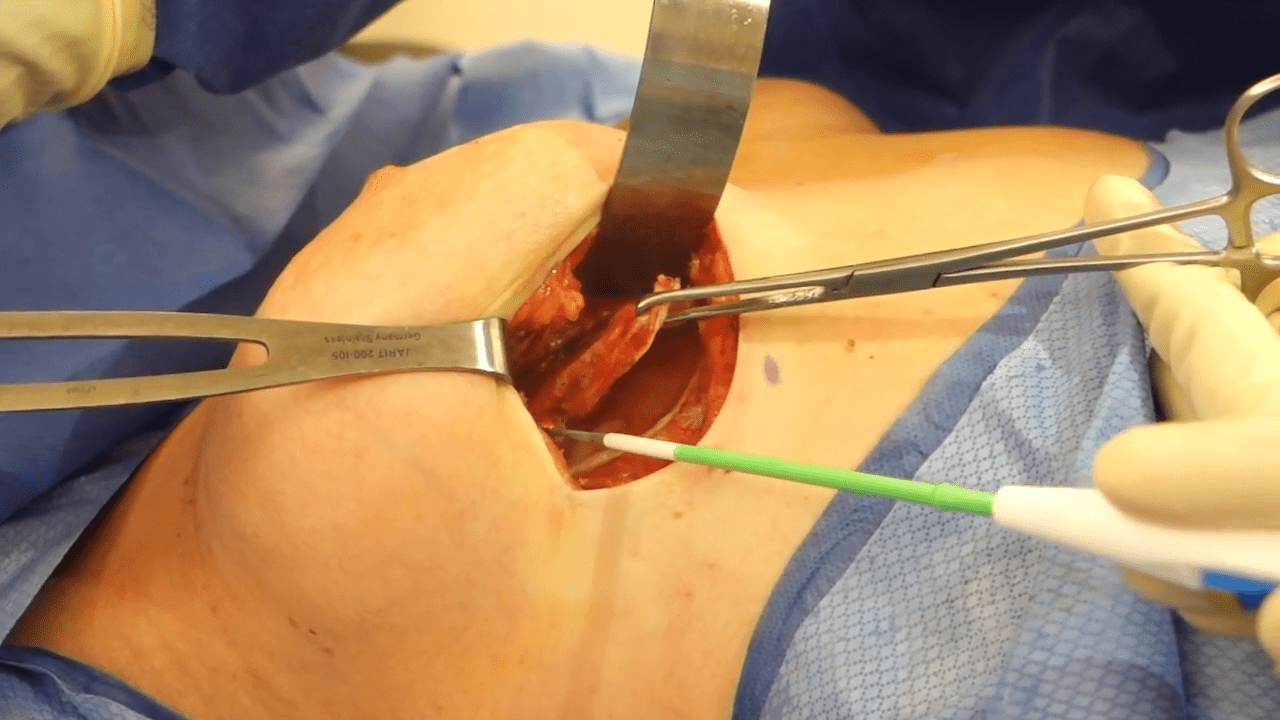
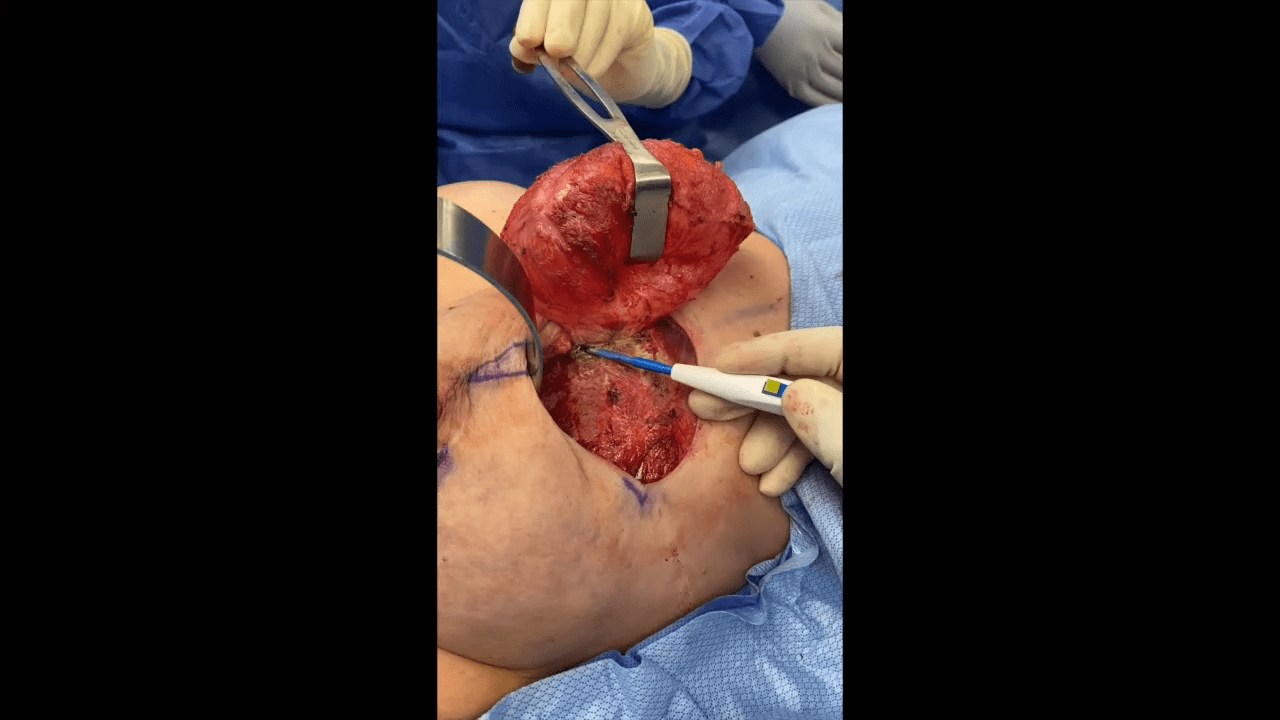
Breast implant removal is a safe, reliable, and outpatient procedure. Prior to breast explantation surgery, there are two important considerations. Breast explantation will change the appearance of the breast, and procedures such as mastopexy (breast lift) and/or fat grafting can be performed at the same time as breast implant removal. Secondly, it is important to determine how the breast capsule will be managed during surgery. With any breast implant, your body creates a layer of protective scar tissue around the implant, called a capsule. The appropriateness as to whether capsule removal is indicated is not always clear and can be quite controversial based on the indication for breast implant removal.